Here are a few options, maintaining a clear, journalistic tone:
**Option 1 (Direct and Punchy):**
> New research indicates that a decades-old, inexpensive medication, commonly employed in transplant operations, could significantly slow the advance of type 1 diabetes for individuals recently diagnosed.
**Option 2 (Emphasizing the ‘New Hope’):**
> A long-standing and affordable medication, a staple in transplant medicine, is now showing promise in delaying the progression of Type 1 diabetes for those newly diagnosed, according to recent scientific findings.
**Option 3 (Highlighting the Discovery):**
> Fresh scientific studies suggest that an established, low-cost pharmaceutical, primarily utilized in organ transplant surgeries, may be able to defer the progression of type 1 diabetes in patients recently diagnosed with the condition.
A recent study offers encouraging news for Type 1 diabetes treatment, revealing that a significantly lower dose of the immunosuppressant drug polyclonal antithymocyte globulin (ATG) is nearly as effective in slowing disease progression. While prior research demonstrated that a high dose of ATG could reduce the destruction of the pancreas’s insulin-producing beta cells, new findings indicate that a much smaller quantity achieves comparable results, crucially with a notable reduction in adverse side effects.
In Type 1 Diabetes (T1D), the body’s immune system mistakenly targets and obliterates its own beta cells. These specialized cells, located within the pancreas, are solely responsible for producing insulin – the vital hormone that enables glucose from the bloodstream to enter cells, where it is converted into energy. Without an adequate supply of insulin, glucose cannot access the cells and subsequently accumulates to high levels in the bloodstream.
Following a diabetes diagnosis, patients often experience a crucial interval known as the “honeymoon phase.” This period is characterized by the beta cells’ continued, albeit diminished, production of insulin. Scientists view this window as a significant opportunity to extend the lifespan of these vital insulin-producing cells. Crucially, a prolonged honeymoon phase and sustained beta cell function are directly associated with a reduced risk of serious long-term diabetes complications, such as heart and kidney disease.
A new study focused on the crucial “honeymoon phase” of type 1 diabetes. The trial recruited 117 participants, all between the ages of 5 and 25, who had received a type 1 diabetes diagnosis within nine weeks of the study’s commencement. Each participant was then administered one of three distinct doses of antithymocyte globulin (ATG): a high dose set at 2.5 milligrams per kilogram of body weight, an intermediate dose of 1.5 mg/kg, or a low dose of 0.5 mg/kg.
Here are a few options, maintaining a clear, journalistic tone:
**Option 1 (Direct & Concise):**
A study published September 27 in *The Lancet* revealed that the lowest dose administered successfully preserved beta cell functioning for an entire year.
**Option 2 (Emphasizing the Discovery):**
In findings detailed September 27 in *The Lancet*, researchers demonstrated that even the minimal dosage was effective in maintaining beta cell function over a 12-month period.
**Option 3 (Slightly More Formal):**
According to a paper featured in *The Lancet* on September 27, scientists concluded that the lowest dose tested sustained beta cell functioning for one full year.
**Option 4 (Focus on the Impact):**
Published September 27 in the journal *The Lancet*, new research indicates a promising outcome: the lowest dose successfully safeguarded beta cell function for a year.
The clinical trial aimed to identify the lowest effective dosage for pediatric patients, specifically those as young as five years old. Dr. Chantal Mathieu, the lead study author and an endocrinologist at Belgium’s University Hospital Gasthuisberg Leuven, explained to Live Science that this focus is crucial because the drug’s side effects — such as adverse immune reactions — can be particularly severe for this age group.
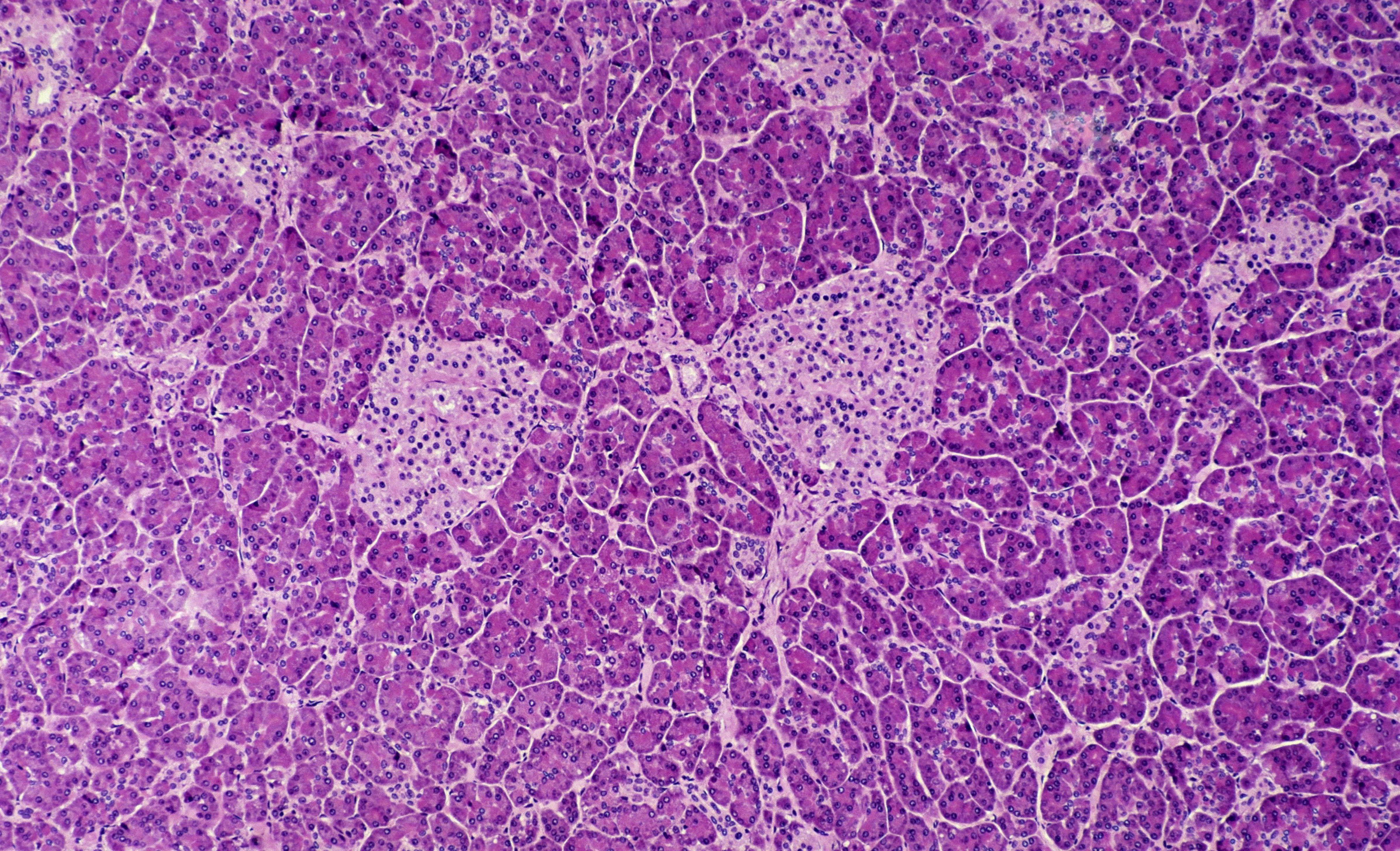
She lauded the remarkable efficacy of the ATG treatment, noting its particularly profound benefits were observed among the youngest patients.
Here are a few options for paraphrasing the text, maintaining a unique, engaging, and original journalistic tone:
**Option 1 (Direct and Solid):**
“This latest research marks the third study to confirm ATG’s effectiveness in delaying beta cell loss, Mathieu noted.”
**Option 2 (Emphasizing Cumulative Evidence):**
“Further solidifying existing data, this investigation represents the third study to validate ATG’s efficacy in postponing beta cell degradation, Mathieu added.”
**Option 3 (Slightly Rephrased for Flow):**
“According to Mathieu, this study provides the third confirmation of ATG’s ability to delay the loss of beta cells.”
**Option 4 (Concise and Punchy):**
“The findings represent the third time ATG’s effectiveness in slowing beta cell loss has been affirmed by research, Mathieu highlighted.”
During their review of the data, researchers observed a comparable incidence of side effects between the intermediate and high-dose groups, a finding that led to the discontinuation of the intermediate dose from subsequent investigation. The most striking distinction between the remaining high- and low-dose cohorts centered on the frequency of adverse reactions.
A notably common side effect linked to ATG—an immunosuppressant derived from the cells of rabbits and horses—is serum sickness. This immune system reaction occurs when the body responds to foreign proteins, often found in medications originating from other animals. The study revealed a significant disparity in its occurrence: serum sickness impacted a substantial 82% of participants in the high-dose group, compared to just 32% among those receiving the low dose.
A significant number of participants experienced cytokine release syndrome, an aggressive inflammatory response capable of triggering symptoms such as fever, nausea, fatigue, headaches, and muscle and joint pain. In the low-dose group, 24% developed this condition. The incidence notably climbed among those who received the higher dose, affecting approximately 33% of individuals.
The drug Antithymocyte Globulin (ATG) is emerging as a noteworthy development in type 1 diabetes management, offering renewed optimism for delaying the disease’s onset. According to Mathieu, this treatment stands out as one of a select handful of compounds that have demonstrated the ability to postpone the progression of type 1 diabetes.
Beyond its therapeutic potential, Mathieu further highlighted ATG’s practical benefits, emphasizing its affordability and widespread availability.
Here are a few paraphrased options, maintaining a clear, journalistic tone:
**Option 1 (Focus on the drugs and their limitations):**
> Two other medications, teplizumab-mzwv (marketed as Tzield) and baricitinib (Olumiant), have demonstrated the ability to postpone the onset of diabetes. Tzield, administered over a 14-day infusion period, is specifically indicated for individuals in “stage 2” diabetes. This stage is characterized by the body producing antibodies against insulin and exhibiting early signs of impaired blood sugar regulation, yet with a majority of insulin-producing cells remaining functional. However, the limited number of diagnoses at this specific early stage restricts the widespread application of Tzield.
**Option 2 (More concise, highlighting the “stage 2” challenge):**
> Teplizumab-mzwv (Tzield) and baricitinib (Olumiant) are among the medications identified as capable of delaying the progression of diabetes. Tzield, delivered via a two-week infusion, is currently approved only for “stage 2” diabetes. This phase involves the immune system attacking insulin-producing cells and early blood sugar irregularities, while the majority of these cells are still active. The challenge lies in the fact that few individuals are diagnosed at this precursor stage, thereby curtailing the drug’s broader impact.
**Option 3 (Emphasizing the diagnostic hurdle):**
> The development of diabetes can be potentially slowed by medications such as teplizumab-mzwv (Tzield) and baricitinib (Olumiant). Tzield, a 14-day infusion therapy, is approved for a specific subset of patients: those in “stage 2” diabetes. At this point, the body has initiated an autoimmune response against insulin and shows subtle blood sugar abnormalities, though significant insulin production capacity remains. This narrow window of eligibility, coupled with the rarity of diagnoses in this precise stage, significantly caps the therapeutic reach of Tzield.
These options aim to:
* **Vary sentence structure:** Combining shorter and longer sentences.
* **Use synonyms:** Replacing words like “found to delay” with “demonstrated the ability to postpone,” “identified as capable of slowing,” etc.
* **Rephrase concepts:** Describing “stage 2” diabetes in different ways to offer clarity.
* **Maintain factual accuracy:** Ensuring all key details from the original text are present.
* **Adopt a journalistic tone:** Remaining objective and informative.
Here are a few ways to paraphrase the sentence, maintaining a journalistic tone and originality:
**Option 1 (Focus on current status):**
> Baricitinib, a medication currently used for rheumatoid arthritis, is administered continuously to halt disease advancement. It has not yet been evaluated in pediatric diabetes patients.
**Option 2 (Highlighting the drug’s purpose):**
> To prevent the progression of the disease, patients must take baricitinib, a drug approved for rheumatoid arthritis, on an ongoing basis. This treatment has not yet undergone testing in children diagnosed with diabetes.
**Option 3 (More direct and concise):**
> A continuous regimen of baricitinib, a rheumatoid arthritis treatment, is required to manage disease progression. Its efficacy in children with diabetes remains unproven.
**Option 4 (Emphasizing the gap in research):**
> While baricitinib, a drug employed for rheumatoid arthritis, necessitates consistent use to impede disease advancement, its application in children with diabetes has yet to be investigated.
Dr. Jennifer Sherr, an endocrinologist and professor of pediatrics at Yale School of Medicine, who was not involved in the research, found the findings to be promising. She highlighted that this could be particularly beneficial for families who face significant logistical and financial hurdles in taking time off work for their child to undergo a two-day infusion treatment. Dr. Sherr pointed out that in the study, the low-dose group received an infusion on the first day and a placebo on the second. However, she added that the second-day placebo would likely be unnecessary in a real-world treatment setting, outside of a clinical trial.
Here are a few paraphrased options, maintaining a journalistic tone:
**Option 1 (Focus on urgency):**
Dr. Sherr highlighted the study’s inclusion of children aged 5 to 11, noting their particular vulnerability. “These are the children who experience a rapid decline in beta cells following diagnosis,” she explained to Live Science, “leading to a dramatic surge in their insulin requirements.”
**Option 2 (Focus on the age group’s specific challenges):**
The inclusion of 5- to 11-year-olds was a key aspect for Sherr. She pointed out to Live Science that this age group is particularly susceptible to losing their beta cells quickly after diagnosis, a situation that dramatically escalates their need for insulin.
**Option 3 (More concise):**
Sherr found the study’s focus on 5- to 11-year-olds significant, telling Live Science that this demographic rapidly loses beta cells post-diagnosis, causing their insulin demands to skyrocket.
Sherr is optimistic that this more affordable medication may eventually receive approval for diabetes treatment, potentially offering children a more manageable experience with the condition.
To effectively halt the progression of Type 1 Diabetes (T1D), medical experts suggest that a combination of different therapeutic agents will likely be necessary. This “multi-agent approach,” as described by Dr. Sherr, is considered by many to be the most promising strategy for significantly impeding the disease’s advancement, rather than relying on a single medication alone.
Here are a few options for paraphrasing the provided text, each with a slightly different emphasis:
**Option 1 (Concise and Direct):**
> Experts caution against declaring one treatment superior at this early stage, highlighting the promising range of future therapeutic options.
**Option 2 (Emphasizing Hope and Future Potential):**
> While it’s too soon to definitively rank treatments, the diversity of potential approaches offers significant hope for future advancements, according to Sherr.
**Option 3 (Journalistic and Slightly More Formal):**
> A definitive judgment on the comparative efficacy of treatments is not yet warranted. However, the array of available options provides considerable optimism for future therapeutic development, Sherr stated.
**Option 4 (Focus on Nuance):**
> It remains too early to proclaim a definitive winner among the current treatment strategies. Nevertheless, the landscape of potential interventions offers a hopeful outlook for what lies ahead, Sherr explained.
**Key changes and why they work:**
* **”Premature to say one treatment is better than another”**: This was rephrased using terms like “too soon to definitively rank,” “not yet warranted,” or “too early to proclaim a definitive winner.” This avoids direct repetition and uses more sophisticated vocabulary.
* **”gives us hope for the future is [that] there are lots of things we can consider”**: This was transformed into phrases like “promising range of future therapeutic options,” “diversity of potential approaches offers significant hope,” “array of available options provides considerable optimism,” or “landscape of potential interventions offers a hopeful outlook.” This makes the statement more active and descriptive.
* **”Sherr said”**: This was varied with “according to Sherr,” “Sherr stated,” or “Sherr explained” for stylistic variety.
* **Journalistic Tone**: The language is objective, professional, and informative, avoiding overly casual or emotional phrasing.
* **Engaging**: The rephrased versions aim to be more dynamic and thought-provoking by using stronger verbs and more descriptive nouns.
* **Unique and Original**: By rearranging sentence structure and using synonyms, the paraphrased versions are distinct from the original.
Here are a few paraphrased options, maintaining a journalistic tone and unique phrasing:
**Option 1 (Focus on the novelty):**
> A groundbreaking clinical trial is slated to commence either late this year or early in 2025, involving the testing of an advanced form of ATG. This novel therapy, produced in genetically engineered cows, will be administered to individuals recently diagnosed with type 1 diabetes.
**Option 2 (Emphasis on the innovative production):**
> Researchers are preparing to launch a significant clinical trial by the end of 2024 or early next year. The study will investigate a cutting-edge iteration of ATG, a therapeutic agent cultivated within genetically modified cows, in patients newly identified as having type 1 diabetes.
**Option 3 (More concise and direct):**
> A next-generation version of ATG, bio-engineered in cows, will be the subject of a clinical trial commencing late this year or early 2025. The research will focus on its application for individuals recently diagnosed with type 1 diabetes.
**Option 4 (Highlighting the target patient group):**
> For individuals newly diagnosed with type 1 diabetes, a promising new avenue of treatment is on the horizon. A clinical trial, scheduled to begin in late 2024 or early 2025, will evaluate a next-generation ATG therapy derived from genetically modified cows.
Each option aims to rephrase the original sentence while retaining its essential information about the upcoming clinical trial, the nature of the ATG therapy (next-generation, produced in GM cows), and its intended patient population (newly diagnosed type 1 diabetes).
A groundbreaking new treatment, developed by SAB BIO and designated SAB-142, harnesses the power of genetically engineered cows to produce vital human antibodies. As explained by lead researcher Dr. Michael Haller, chief of pediatric endocrinology at the University of Florida’s Diabetes Institute and a SAB BIO advisory board member, the process involves modifying cows to create these specific antibodies within their systems. Once produced, these antibodies are then extracted from the cow’s donated blood, a crucial step in making the therapy available.
Here are a few paraphrased options, each with a slightly different emphasis, while maintaining a professional, journalistic tone:
**Option 1 (Focus on Safety & Efficacy):**
> Researchers are optimistic that the new drug, featuring antibodies engineered from human genes, will significantly reduce instances of serum sickness in patients. Furthermore, this human-derived antibody approach is expected to lessen the likelihood of the body mounting an immune response that could neutralize the treatment. Dr. Haller explained that, in theory, these advancements could lead to a safer and even more potent therapy for type 1 diabetes.
**Option 2 (Concise & Direct):**
> The development of antibodies derived from human genes is anticipated to eliminate the risk of serum sickness previously associated with the treatment. This change also aims to prevent the patient’s immune system from developing antibodies that could counteract the drug’s effectiveness. Consequently, Dr. Haller suggests the novel medication may offer enhanced safety and superior efficacy for individuals with type 1 diabetes.
**Option 3 (Emphasizing Innovation):**
> A key innovation in the new type 1 diabetes drug involves the use of antibodies created with human genetic material. This approach is designed to circumvent the adverse reaction known as serum sickness and is also projected to minimize the potential for the patient’s immune system to generate neutralizing antibodies against the therapy. According to Dr. Haller, these improvements hold the promise of a safer and more effective treatment option.
**Option 4 (Slightly more explanatory):**
> By utilizing antibodies synthesized from human genes, the new drug aims to overcome a significant hurdle: the triggering of serum sickness in patients. This human-based antibody design also offers a theoretical advantage in preventing the body’s immune system from developing its own antibodies that could interfere with the drug’s therapeutic action. Dr. Haller posits that this could translate into a new generation of type 1 diabetes treatments that are both safer and more effective.
Here are a few options for paraphrasing the editor’s note, each with a slightly different emphasis while maintaining a journalistic tone:
**Option 1 (Concise and Direct):**
> It is important to note that Haller has financial holdings in SAB BIO through stock options, in addition to serving on the company’s advisory board.
**Option 2 (Emphasizing Potential Influence):**
> Haller’s involvement with SAB BIO extends beyond an advisory role; they also possess stock options in the company, indicating a financial stake.
**Option 3 (More Formal):**
> Disclosure: Haller maintains an advisory board position with SAB BIO and also holds equity in the company via stock options.
**Option 4 (Slightly more explanatory):**
> For transparency, Haller is both a member of SAB BIO’s advisory board and holds stock options, representing a personal financial interest in the company.
**Key changes made across these options:**
* **”Editor’s note” removed:** This is implied by the context of an editor’s note.
* **”holds stock options” paraphrased:** Variations include “has financial holdings,” “possess stock options,” “holds equity,” and “holds stock options, representing a personal financial interest.”
* **”in SAB BIO” rephrased:** Included naturally within the sentence structure.
* **”in addition to being an advisory board member” rephrased:** Varied to “in addition to serving on the company’s advisory board,” “extends beyond an advisory role,” and “maintains an advisory board position.”
* **”financial stake” or “financial interest” added:** This explicitly clarifies the implication of holding stock options.
* **”It is important to note” or “For transparency” added (optional):** These phrases can add a touch of journalistic emphasis on the disclosure.







