Two pioneering scientists have been awarded the prestigious $3 million Breakthrough Prize in Life Sciences for their groundbreaking contributions that led to the development of the first approved therapy utilizing the revolutionary CRISPR gene-editing technology.
Distinguished researchers Dr. Swee Lay Thein, from the National Heart, Lung and Blood Institute (NHLBI), and Dr. Stuart H. Orkin, of Harvard University, have been jointly honored with an award for their foundational scientific work. Their pioneering basic research culminated in the development of a transformative gene therapy designed to treat the severe blood disorders, sickle cell disease and beta-thalassemia.
Casgevy, a groundbreaking new treatment, offers a functional cure for debilitating and often fatal diseases by precisely disabling a single gene. Scientists Thein and Orkin were formally recognized for their contributions, accepting their awards at a ceremony held in Los Angeles on Saturday, April 18.
Here are a few options, each with a slightly different emphasis, while maintaining the core meaning:
**Option 1 (Concise and direct):**
Speaking to Live Science, Thein conveyed a profound sense of honor, admitting he felt both overwhelmed and deeply humbled by the experience.
**Option 2 (Emphasizing the mix of emotions):**
In an interview with Live Science, Thein described his reaction as a powerful blend of emotions, stating he was “extremely honored, overwhelmed, and humbled.”
**Option 3 (Focusing on the impact):**
Thein shared with Live Science that the situation had left him feeling immensely honored, emotionally overwhelmed, and profoundly humbled.
**Option 4 (Slightly more formal):**
According to Thein, who spoke with Live Science, the circumstances elicited a strong emotional response, prompting feelings of great honor, an overwhelming sensation, and a deep sense of humility.
Since its inception in 2013, the esteemed Breakthrough Prize in Life Sciences has been dedicated to honoring groundbreaking discoveries and significant advancements across the breadth of the life sciences.
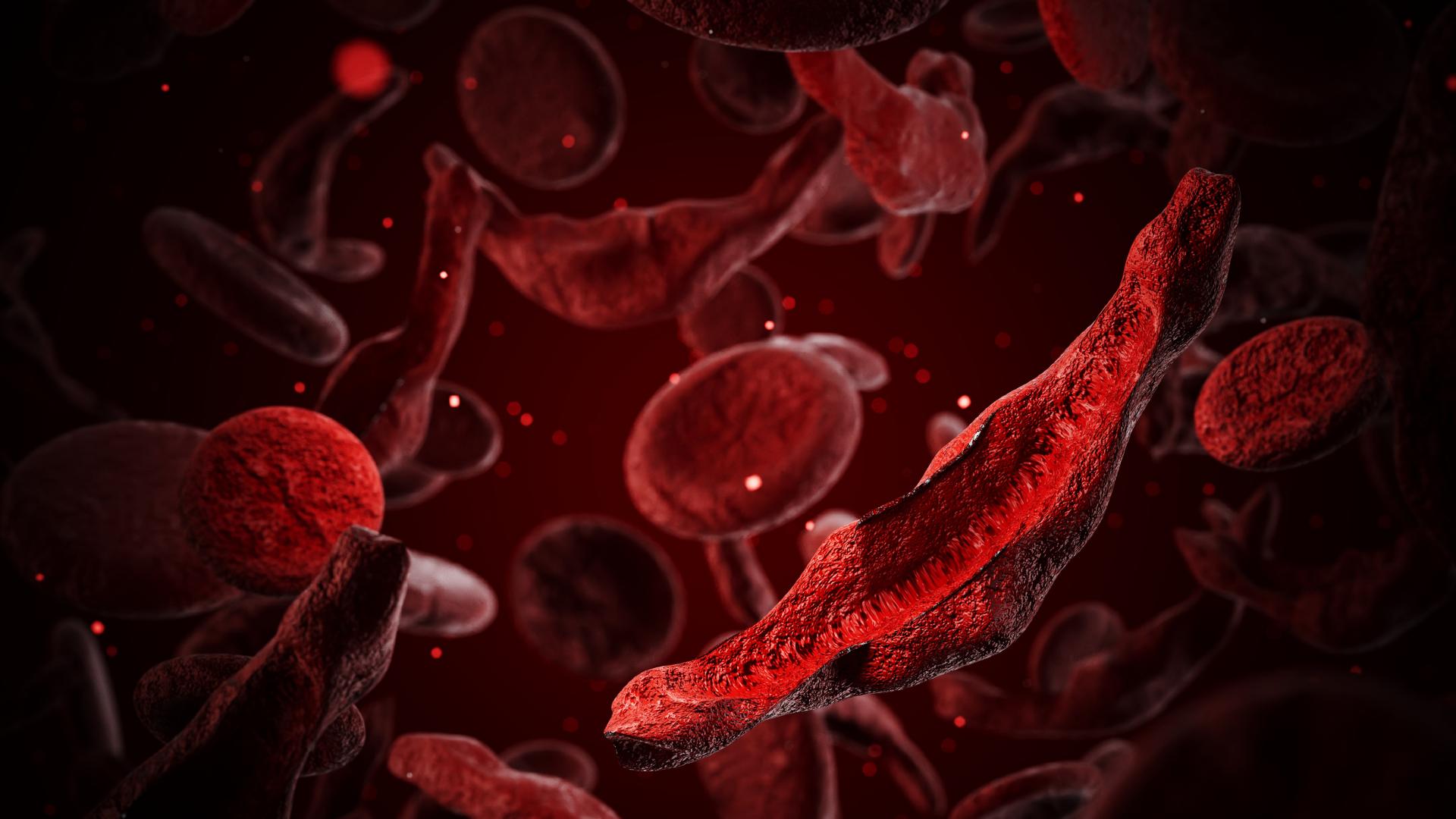
Sickle cell disease, a significant global health challenge, affects an estimated 7 to 8 million individuals worldwide, with the vast majority of cases concentrated in Africa. The defining characteristic of this genetic disorder is the abnormal shape of red blood cells, which transform into rigid, crescent-like structures. This deformation occurs because hemoglobin, the vital oxygen-carrying molecule within these cells, forms stiff, elongated fibers.
These malformed “sickled” cells pose several severe health risks. They are prone to adhering to one another, leading to dangerous blood clots that can obstruct blood flow. Moreover, their inherent fragility makes them susceptible to premature bursting and death, resulting in chronically low red blood cell counts, a condition known as anemia.
Sickle cell patients frequently endure agonizing pain episodes, termed “crises,” when abnormally shaped red blood cells obstruct crucial blood vessels. These debilitating blockages don’t just cause pain; they can inflict severe damage on vital organs like the lungs, liver, and spleen. Of particular concern, obstructions in the lungs can precipitate “acute chest syndrome,” a life-threatening complication that critically depletes oxygen levels and stands as the leading cause of mortality among sickle cell sufferers.
Beta-thalassemia is a severe genetic blood disorder marked by the body’s inability to produce adequate amounts of beta-globin, a crucial component of the hemoglobin molecule. This deficiency means that individuals afflicted with the severe forms of the disease are dependent on lifelong blood transfusions to survive. In a significant development, Casgevy has now received approval as a treatment for these critical manifestations of beta-thalassemia.
Here are a few options for paraphrasing the sentence, each with a slightly different emphasis:
**Option 1 (Focus on the “why”):**
> Dr. Thein, a senior investigator at the NHLBI, embarked on her research in the 1980s, seeking to unravel the mystery behind why certain individuals experienced significantly less severe forms of these particular disorders.
**Option 2 (Focus on the “investigation”):**
> In the 1980s, Dr. Thein, now a senior investigator at the NHLBI, initiated her scientific pursuit to understand the underlying reasons for the milder manifestations of these diseases observed in some patients.
**Option 3 (More direct and concise):**
> Senior NHLBI investigator Dr. Thein’s work, which started in the 1980s, focused on identifying the factors that contributed to less severe cases of these disorders in some individuals.
**Option 4 (Emphasizing the “variation”):**
> Back in the 1980s, Dr. Thein, a senior investigator at the NHLBI, commenced her research aimed at explaining the considerable variation in disease severity seen among individuals with these specific conditions.
**Key changes made across these options:**
* **”began her work”** replaced with: “embarked on her research,” “initiated her scientific pursuit,” “focused on,” “commenced her research.”
* **”trying to figure out why”** replaced with: “seeking to unravel the mystery behind why,” “to understand the underlying reasons for,” “identifying the factors that contributed to,” “aimed at explaining.”
* **”some people… had much milder forms of the diseases than others”** replaced with: “certain individuals experienced significantly less severe forms of these particular disorders,” “the milder manifestations of these diseases observed in some patients,” “less severe cases of these disorders in some individuals,” “the considerable variation in disease severity seen among individuals with these specific conditions.”
* **Added “Dr.”** to Thein for professional tone.
* **”NHLBI”** kept as an acronym as it’s likely established context.
Choose the option that best fits the flow and tone of the surrounding text.
Decades prior, pediatrician Dr. Janet Watson in New York observed a critical insight: infants who would later be diagnosed with sickle cell disease exhibited no outward symptoms and possessed red blood cells that did not undergo sickling.
As children transitioned into toddlerhood, the observable signs of the illness began to manifest.
New research has revealed a fascinating shift in human development, with the body producing distinct types of hemoglobin at different life stages. Before birth, a form known as “fetal hemoglobin” is prevalent in the womb. However, as infants grow and mature, production of this initial type gradually ceases, giving way to the dominant “adult hemoglobin.”

Here are a few options for paraphrasing the quote, each with a slightly different emphasis:
**Option 1 (Focus on the scientific pursuit):**
> Dr. Thein explained to Live Science that his research began with gathering families affected by mild thalassemia. His primary goal was to decipher the genetic underpinnings of the condition, driven by the clear observation that these individuals possess an inherent capacity to sustain the production of fetal hemoglobin.
**Option 2 (More active and direct):**
> “I began by identifying families with mild thalassemia, aiming to uncover the genetic basis of their condition,” Dr. Thein shared with Live Science. He noted that it became apparent they possessed a natural, innate ability to keep producing fetal hemoglobin.
**Option 3 (Emphasizing the “why”):**
> Driven by a desire to understand the genetic roots of mild thalassemia, Dr. Thein started compiling data from affected families, as he told Live Science. He found it evident that these patients demonstrated a natural, inborn talent for continuing to produce fetal hemoglobin.
**Option 4 (Concise and informative):**
> According to Dr. Thein’s interview with Live Science, his initial research focused on collecting data from families with mild thalassemia to unravel its genetics. He observed a distinct, natural ability in these patients to continue producing fetal hemoglobin.
Each of these options rephrases the original quote while retaining the key information about:
* **The researcher’s name:** Thein
* **The subject of study:** Mild thalassemia
* **The research objective:** Unraveling the genetics
* **The key observation:** Innate ability to produce fetal hemoglobin
Here are a few options for paraphrasing the sentence, each with a slightly different emphasis:
**Option 1 (Focus on scope and depth):**
> The researcher delved into the genetic makeup of multiple families afflicted by hereditary diseases. Notably, her study encompassed an extensive Indian family lineage, tracing over 200 members across seven generations and spanning diverse geographical locations worldwide.
**Option 2 (More direct and concise):**
> Her genetic analysis focused on families with a history of disease, prominently featuring a sprawling Indian family with over 200 members documented across seven generations and multiple continents.
**Option 3 (Emphasizing the generational and geographical reach):**
> Investigating the genetic underpinnings of disease, she examined several family lines. A significant part of this research involved a remarkable Indian family, whose lineage of more than 200 individuals extended through seven generations and was dispersed across various continents.
**Option 4 (Slightly more narrative):**
> To understand the genetic transmission of disease, she studied several families. Among them was a vast Indian family tree, boasting over 200 members, a history stretching back seven generations, and members residing on different continents.
**Key changes made in these paraphrases:**
* **”analyzed the genes of”** replaced with phrases like “delved into the genetic makeup of,” “genetic analysis focused on,” “examined the genetic underpinnings of,” “studied the genetic transmission of.”
* **”several families that had a history of disease”** rephrased as “multiple families afflicted by hereditary diseases,” “families with a history of disease,” “family lines with a history of disease,” “families with a history of disease.”
* **”including a family of Indian origin”** became “Notably, her study encompassed an extensive Indian family lineage,” “prominently featuring a sprawling Indian family,” “A significant part of this research involved a remarkable Indian family,” “Among them was a vast Indian family tree.”
* **”that included more than 200 members, spanned seven generations and lived on multiple continents”** was reordered and rephrased for flow and variety, using terms like “tracing over 200 members across seven generations and spanning diverse geographical locations worldwide,” “with over 200 members documented across seven generations and multiple continents,” “whose lineage of more than 200 individuals extended through seven generations and was dispersed across various continents,” “boasting over 200 members, a history stretching back seven generations, and members residing on different continents.”
* **Tone:** Maintained a professional, journalistic, and informative tone.
* **Originality:** Used different vocabulary and sentence structures to avoid direct copying.
Researchers, led by Thein, gained a pivotal understanding by examining twin pairs—both identical and fraternal—who exhibited exceptionally high or low concentrations of fetal hemoglobin. This groundbreaking study allowed the team to pinpoint specific gene variations that influence the body’s production of this crucial blood component. Their investigation ultimately focused on a particular area of the BCL11A gene, located on chromosome 11.
Researchers have identified a specific gene, BCL11A, that acts as a switch to turn off the production of fetal hemoglobin shortly after birth. However, individuals possessing certain variations of this gene find that the “off” switch malfunctions, allowing fetal hemoglobin to remain at high levels throughout their lives.
Building on this foundation, the idea that inhibiting the inhibitor offered a promising therapeutic avenue for individuals suffering from severe forms of sickle cell disease or beta-thalassemia emerged as a logical next step. The groundbreaking work of Orkin was instrumental in validating this crucial conceptual shift.
Dr. Orkin, a leading pediatric hematologist and oncologist affiliated with Boston Children’s Hospital, Dana-Farber Cancer Institute, Harvard Medical School, and the Howard Hughes Medical Institute, has elucidated the mechanism by which a specific repressor protein facilitates the transition to adult hemoglobin. His research also demonstrates that gene editing techniques can be employed to target this crucial genetic region.
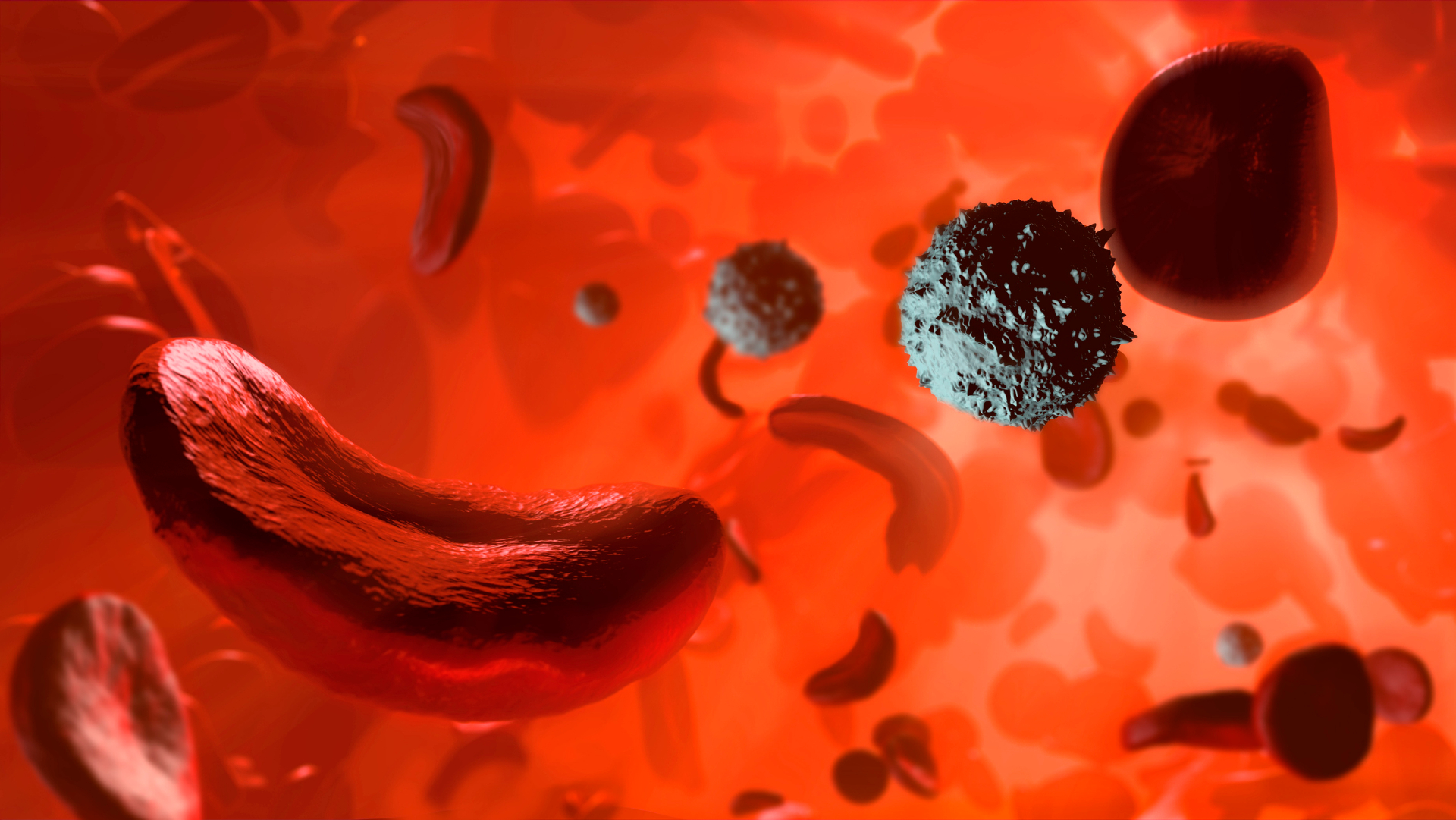
Vertex Pharmaceuticals employed the revolutionary CRISPR gene-editing technology, akin to a molecular scissors, to excise the BCL11A gene’s repressor segment.
The innovative gene therapy, Casgevy, is the culmination of extensive research and development. Administering this groundbreaking treatment involves a precise, multi-step process: initially, a patient’s bone marrow cells are extracted. These cells are then genetically edited outside the body using CRISPR technology, specifically targeting and modifying the BCL11A repressor. Following this crucial genetic alteration, the engineered bone marrow cells are reinfused back into the patient, where they subsequently begin to produce red blood cells rich in fetal hemoglobin.

Heralded as the first “functional cure” for sickle cell disease, this transformative therapy has already revolutionized the lives of the few patients fortunate enough to receive it. Yet, as Dr. Thein notes, its widespread application faces substantial hurdles, accompanied by significant drawbacks.
The comprehensive treatment itself is a demanding endeavor, often spanning up to an entire year to complete. Financially, it carries an astonishing price tag, frequently reaching into the multi-million dollar range. Furthermore, patients must undergo a rigorous and often debilitating course of chemotherapy, a critical step required to clear out existing bone marrow and create the necessary space for the gene-edited stem cells to successfully engraft.
Thein underscored the profound physical demands placed upon patients.
Sickle cell disease and beta-thalassemia disproportionately impact populations across Africa, Asia, and the Mediterranean, often in regions where the essential medical infrastructure and specialized resources required for such treatments are scarce. Recognizing this critical challenge, gene therapy scientists are now prioritizing an “in vivo” approach. This innovative method, as explained by Thein, involves directly injecting the gene-editing machinery into a patient’s body, thus circumventing the complex and resource-intensive process of extracting, modifying, and reinfusing bone marrow cells.
The imperative for advancements in drug development, especially for therapies that are both more cost-effective and simpler to administer — be they oral medications, injectables, or IV treatments — continues to be a crucial concern, Thein highlighted.
Research led by Thein has focused on Mitavipat, a drug that appears to bolster the metabolic health of red blood cells. According to Thein, this medication is already approved for treating two specific blood disorders: pyruvate kinase deficiency and beta thalassemia.
For patients with certain conditions, one medication has proven to be a long-term game-changer, with one physician noting, “Some of the patients on this drug have been on this treatment and with me for six years, and it has really made quite a big difference.” However, before this promising treatment can be made available to individuals living with sickle cell disease, additional clinical trials are required for regulatory approval.